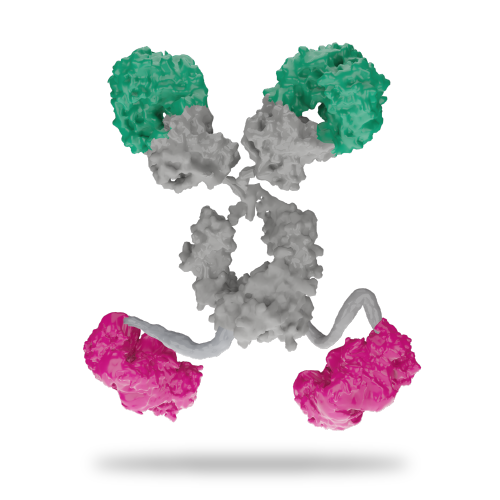
SI-B003
By combining bi-valency with bi-specificity in the tetravalent format, the dual checkpoint molecule utilizes avidity and bi-specificity to improve anti-cancer immune cell function. The specificity enhancement both synergistically enhance and expand the breadth of immune cell activity that is diminished in cancer patients (SEBA). The spatio-temporal control of bi-specificity ensure that synergistic blocking of inhibitory receptors is happening in the right place for greatest effect.
The tetravalent SI-B003 has two binding domains for T cell checkpoint axis, PD-1 and CTLA-4. The primary targets for this molecule are exhausted tumor-specific T cells which demonstrate enhanced functionality upon treatment with PD-1 and CTLA-4 blocking antibodies, restoring their anti-tumor activity.
SI-B003 is currently being evaluated as a single agent in clinical trials enrolling patients with a variety of solid tumor indications.
References
https://ClinicalTrials.gov/show/NCT04606472
Phase II
Phase I
IND Enabling
Pre-Clinical
Platform
Indication
Project
Solid Tumor
SEBA
SI-B003
CTLA-4xPD-1
Pipeline Exploration